© 2025 MJH Life Sciences™ , Patient Care Online – Primary Care News and Clinical Resources. All rights reserved.
15 Drugs Approved for Primary Care in 2022
With 2022 at an end, Patient Care Online has compiled a list of key drugs approved by the US Food and Drug Administration (FDA) for conditions that are often seen in primary care. Scroll through the slides below to find details on a new oral solution of amlodipine; the first and only FDA-approved, once-daily amphetamine transdermal patch for the treatment of attention deficit hyperactivity disorder; the first and only specific interleukin-23 inhibitor to be approved for the treatment of Crohn disease in adults; the first drug indicated for the treatment for eosinophilic esophagitis; a novel rapid-acting treatment for major depressive disorder; and 10 more.

Insomnia: Daridorexant tablets (Quviviq®), an orexin receptor antagonist indicated for the treatment of adults with insomnia characterized by difficulties with sleep onset and/or sleep maintenance. The recommended dosage is 25 mg to 50 mg once per night, taken orally within 30 minutes before going to bed, with at least 7 hours remaining prior to planned awakening. It is anticipated to be available to patients in May 2022, pending controlled substance scheduling by the FDA. (Approved 1/7/2022, Idorsia Ltd.)
For full prescribing information, please click here.
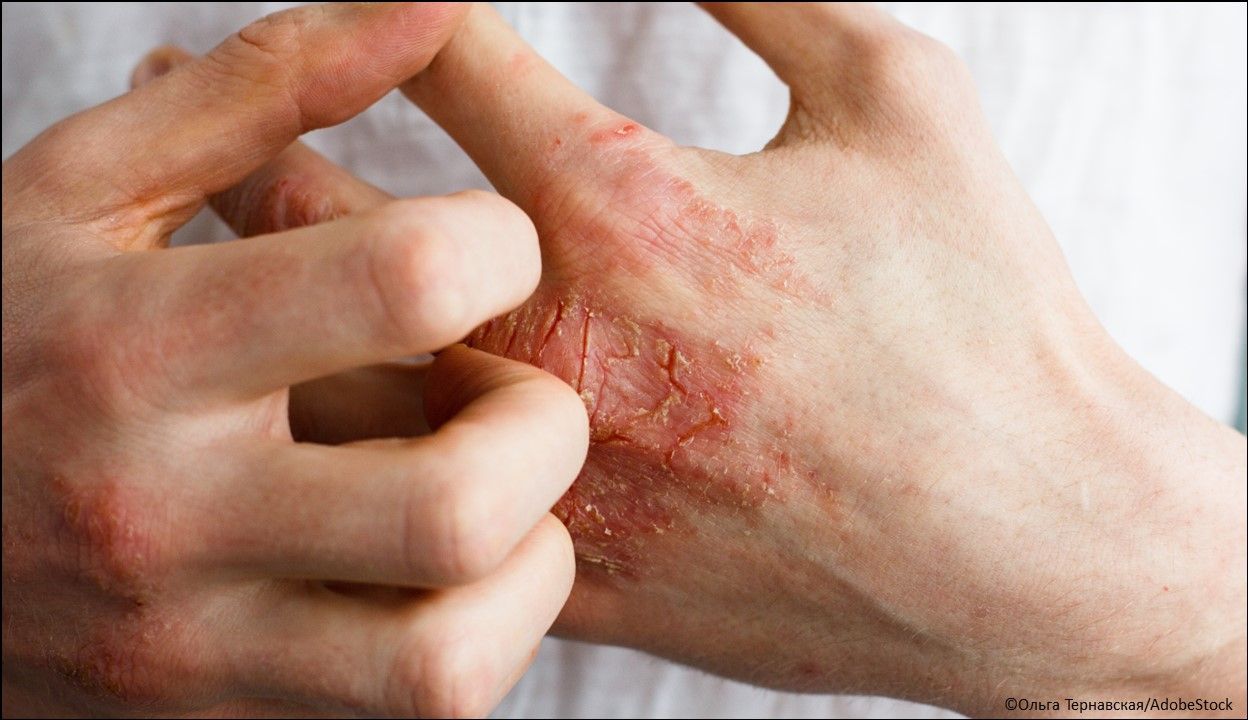
Atopic dermatitis: Abrocitinib tablets (Cibinqo®), a Janus kinase (JAK) inhibitor indicated for the treatment of adults with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable. It is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or immunosuppressants. (Approved 1/14/2022; Pfizer Inc)
For full prescribing information, please click here.
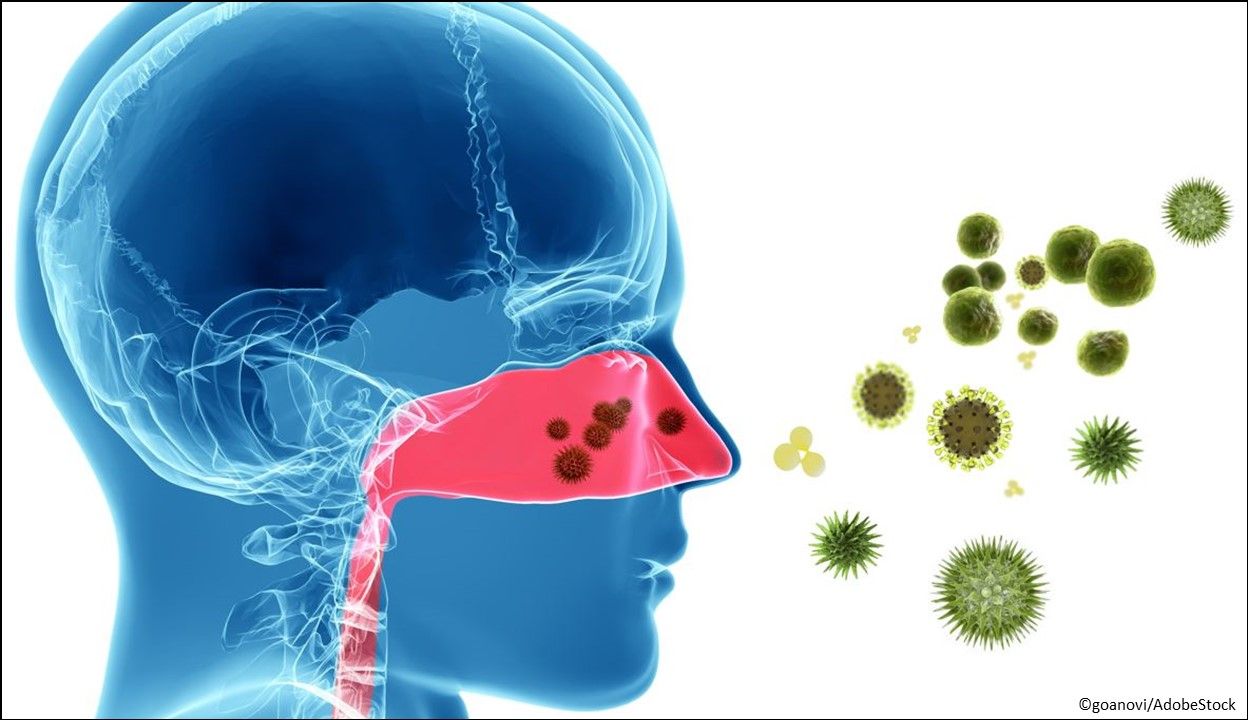
Allergic rhinitis: Olopatadine hydrochloride and mometasone furoate nasal spray (Ryaltris®), a combination of olopatadine, a histamine-1 receptor inhibitor, and mometasone furoate, a corticosteroid, indicated for the treatment of symptoms of seasonal allergic rhinitis in patients aged ≥12 years. The recommended dosage is 2 sprays per nostril twice daily. (Approved 1/13/2022, Glenmark Pharmaceuticals, Inc.)
For full prescribing information, please click here.
COVID-19 prevention: Spikevax (formerly mRNA-1273), a vaccine indicated for active immunization to prevent COVID-19 caused by SARS-CoV-2 in adults aged ≥18 years. Spikevax is administered intramuscularly as a series of 2 doses (0.5 mL each) 1 month apart. (Approved 1/31/2022, Moderna, Inc.)
For the full prescribing information, please click here.
Cardiovascular diseases: Amlodipine oral solution (Norliqva®), 1 mg/mL, a calcium channel blocker indicated for the treatment of hypertension in patients aged ≥6 years, and coronary artery disease (chronic stable angina, vasospastic angina, and angiographically documented coronary artery disease) in patients without heart failure or an ejection fraction <40%. Norliqva is supplied as a peppermint flavored oral solution and is administered once daily. (Approved 1/24/2022, CMP Pharma Inc.)
For full prescribing information, please click here.
Allergic rhinitis: Mometasone furoate monohydrate 50 mcg (Nasonex® 24HR Allergy), a corticosteroid nasal spray, was FDA-approved for over-the-counter use for the temporary relief of the symptoms of hay fever or other upper respiratory allergies. (Approved 3/17/2022; Perrigo Company plc)
For the product label, please click here.
Type 2 diabetes (T2D): Tirzepatide injection (Mounjaro™), a glucose-dependent insulinotropic polypeptide receptor and glucagon-like peptide-1 receptor agonist indicated as an adjunct to diet and exercise to improve glycemic control in adults with T2D. Tirzepatide has not been studied in patients with a history of pancreatitis and is not indicated for use in patients with type 1 diabetes mellitus. The recommended starting dosage is 2.5 mg injected subcutaneously once weekly, and after 4 weeks, increase to 5 mg. (Approved 5/13/2022, Eli Lilly and Company)
For full prescribing information, please click here.
Hypertrophic cardiomyopathy (HCM): Mavacamten capsules (Camzyos™), a first-in-class cardiac myosin inhibitor indicated for the treatment of adults with symptomatic class II-III obstructive HCM to improve functional capacity and symptoms. It is the only treatment that specifically targets the pathophysiology of the disease. (Approved 4/28/2022, Bristol Myers Squibb)
For full prescribing information, please click here.
Measles, mumps, and rubella: Measles, Mumps and Rubella Vaccine, Live (Priorix®), is indicated for active immunization for the prevention of measles, mumps, and rubella in individuals aged ≥12 months. Each dose is approximately 0.5 mL, and it is recommended that the first dose be administered between 12 and 15 months of age and the second dose be administered at 4 through 6 years of age. (Approved 6/3/2022, GSK)
For the package insert, please click here.
Crohn disease (CD): Risankizumab-rzaa (Skyrizi®), is the first and only specific interleukin-23 (IL-23) inhibitor to be approved for the treatment of moderate-to-severe active CD in adults. The dosing regimen for adults with CD is 600 mg administered by intravenous infusion over at least 1 hour at week 0, week 4, and week 8, followed by 360 mg self-administered by subcutaneous injection at week 12, and every 8 weeks thereafter. (Approved 6/16/2022, AbbVie, Inc.)
For full prescribing information, please click here.
Plaque psoriasis: Tapinarof cream (Vtama®) 1%, is a topical aryl hydrocarbon receptor modulating agent indicated for the treatment of plaque psoriasis in adults. Most common adverse reactions associated with tapinarof include follicultitis, nasopharyngitis, contact dermatitis, headache, pruritus, and influenza. (Approved 5/23/22; Dermavant Sciences)
For full prescribing information, please click here.

Major depressive disorder (MDD): Dextromethorphan and bupropion extended-release tablets (Auvelity®) (formerly AXS-05), is the first and only oral N-methyl D-aspartate (NMDA) receptor antagonist approved for the treatment of MDD in adults and uses the first new oral nonmonoamine-based mechanism of action approved in more than 60 years. It is also the first and only rapid-acting medicine approved to treat MDD labeled with statistically significant antidepressant efficacy vs placebo starting at 1 week. (Approved 8/18/2022; Axsome Therapeutics, Inc.)
For full prescribing information, please click here.
Heart failure (HF): Furosemide injection (FUROSCIX®), 80 mg/10 mL, is a loop diuretic indicated for the at-home treatment of congestion due to fluid overload in adults with NYHA Class II/III chronic HF. It is not indicated for emergency situations or in patients with acute pulmonary edema. (Approved 10/07/22, scPharmaceuticals, Inc.)
For full prescribing information, please click here.
Prevention of recurrent Clostridioides difficile infections (rCDI): RBX2660 (Rebyota®), 150 mL, is the first-ever microbiota-based live biotherapeutic indicated for the prevention of rCDI in adults who have completed a course of antibiotic treatment for rCDI. It is prepared from stool donated by qualified healthy individuals and comes as a prepackaged single dose that is administered rectally. It is not indicated for treatment of CDI. (Approved 11/30/22; Ferring Pharmaceuticals Inc.)
For full prescribing information, please click here.
Delaying the Onset of Stage 3 Type 1 Diabetes (T1D): Teplizumab-mzwv Injection (Tzield®), 2 mg/2 mL, a CD3-directed antibody indicated to delay the onset of Stage 3 T1D in adults and pediatric patients aged 8 years and older with Stage 2 T1D. (Approved 11/17/22; Provention Bio, Inc.)
For full prescribing information, please click here.




